NOTE: This analysis was updated in April, 2021.
Two major legislative efforts passed at the onset of the pandemic in March 2020 – the Families First Coronavirus Response Act (FFCRA) and the Coronavirus Aid, Relief, and Economic Security (CARES) Act – required health coverage for COVID-19 testing – including the test itself, the related visit, and other services related to testing – with no cost-sharing for people covered by most private health plans, Medicare, and Medicaid. Federal laws also made resources available to finance free testing for uninsured individuals.
Since enactment, questions have arisen about the scope of protections and when they apply. For example, some plans denied COVID-19 testing claims or applied cost sharing for COVID-19 testing for asymptomatic individuals unless they had known or suspected exposed to COVID-19 and had a referral for testing from their provider. Some denied COVID-19 testing claims unless directly ordered by a physician.
Federal guidance released in early 2021 under the Biden Administration clarified that insurers must cover testing without cost sharing for asymptomatic individuals and without requiring medical screenings. However, insurers are not required to cover COVID-19 testing without cost-sharing if it is conducted as part of employee return-to-work programs or public health surveillance purposes. Such limits mean some patients with health coverage may nonetheless receive bills for COVID-19 diagnostic testing and related services, and those bills often can be widely different from patient to patient.
Outside of the Medicare program, there is no federal regulation of the price of COVID-19 diagnostic tests or other related tests and visits. Much like for other health services, hospitals and laboratories can set their own rates for privately insured and uninsured individuals. Private health insurers negotiate allowed charges with hospitals and providers participating in their network. In the absence of a negotiated rate for out-of-network providers, insurers must pay the provider’s cash price – sometimes called the list price – for COVID-19 testing and related services. The CARES Act requires providers to post the cash price on their public website for insurer use. These CARES Act coverage and payment requirements apply during the duration of the public health emergency.
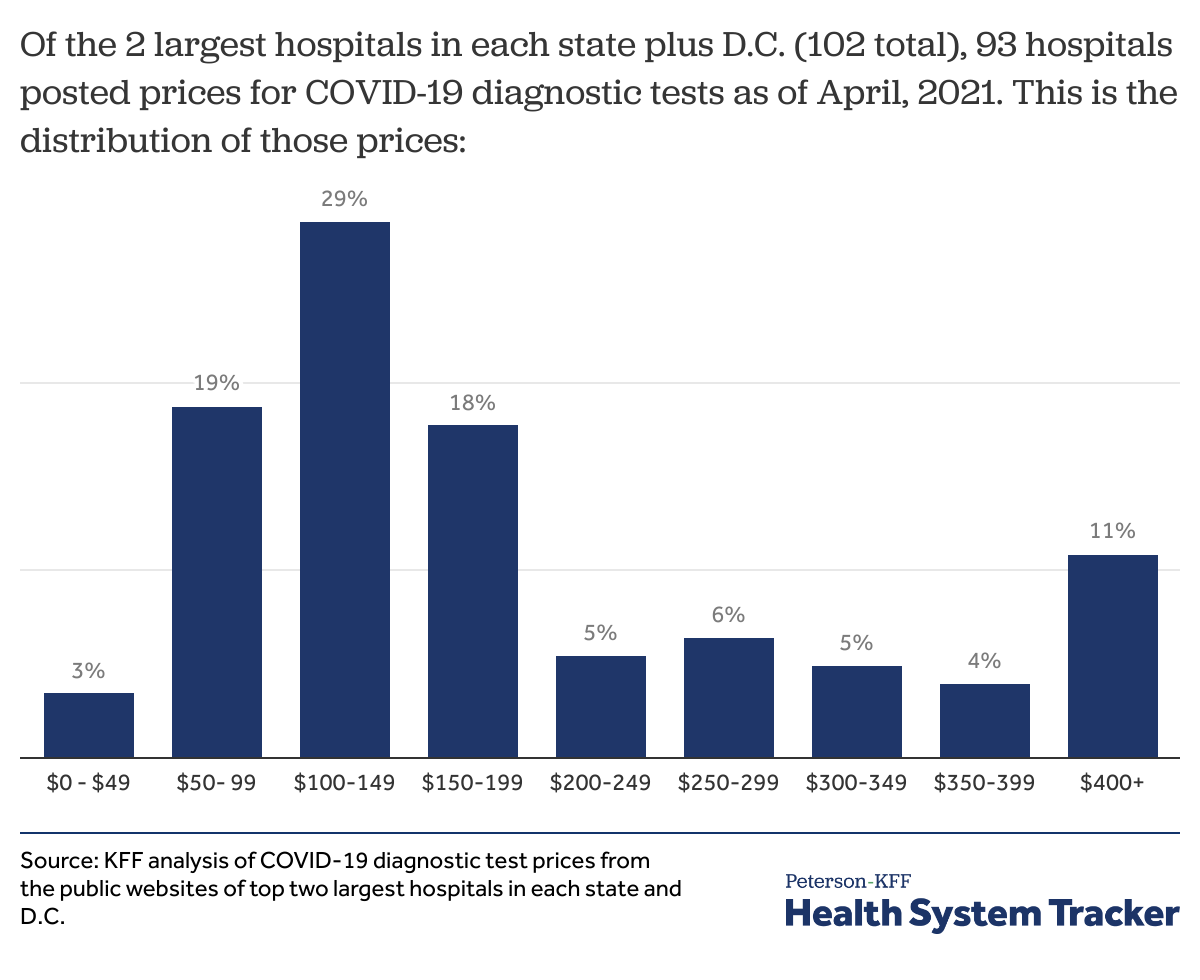
Negotiated rates (between payers and their in-network providers for COVID-19 testing) are not always publicly available. However, the CARES Act price transparency rule provides a unique opportunity to examine what insurers pay for out-of-network care that is required to be covered. In this brief, we present the findings from our search for COVID-19 test prices, which we conducted from April 20 through April 27, 2021. For each state, we searched for COVID-19 test prices posted on public websites of the two largest hospitals. We were able to find COVID-19 diagnostic test prices for 93 out of 102 hospitals, ranging from $20 – $1,419 per single test, not including the price of a provider visit, facility fee, or specimen collection. These services are required to be covered by insurance; nonetheless patients may experience problems with billing or claims denials. Some patients may be billed for testing-related care from the hospital, a provider, or the insurer. We examine federal requirements and guidance on who can be charged for COVID-19 testing and related services. We then end with a broad discussion on how the uncertainty around what patients will be charged for health care services interacts with federal attempts at price transparency.
Hospital list prices for COVID-19 tests vary widely
Medicare covers testing without cost-sharing for patients, and reimburses providers between $36 to $143 per diagnostic test, depending on the type of test and how quickly the test is processed. However, since there are no regulations on pricing in the private market, the price insurers must pay in-network hospitals and providers under current law is their established and negotiated rate for the service. The CARES Act requires insurers to reimburse out-of-network providers the cash price of COVID-19 tests posted on the provider’s website, though they may choose to bargain that price down on a claim-by-claim basis.
We searched the websites of the two largest hospitals in each state and the District of Columbia to see whether the cash price for COVID-19 diagnostic tests were available, and how much those prices varied (see Methods). We found price information for COVID-19 diagnostic tests online for 93 out of the 102 hospital websites we examined. Prices for COVID-19 tests presented in this analysis do not include the price of related services, such as COVID-19 screenings conducted in-office or through telemedicine, or for specimen collection; however, some hospitals also post pricing information for these. Some COVID diagnostic tests may be billed as a panel and include detection for other viruses as well, such as influenza. COVID diagnostic tests that include testing for influenza and respiratory syncytial virus are included in this analysis.
We observed a broad range of COVID-19 testing prices, even within the same hospital system. A given hospital may choose to post different prices for a test for a number of reasons, including the type of test, where it was conducted, and which laboratory analyzes the medical sample.
Prices for COVID-19 diagnostic tests vary across the largest hospitals in each state
Confirmation of an active SARS-CoV-2 infection (the virus that causes COVID-19) is done via tests that use molecular “PCR” amplification and antigen-based lab technology. Data from 93 hospitals with listed prices yielded 203 distinct prices for diagnostic tests. The prices ranged between $20 – $1,419 per diagnostic test, with a median of $148. Nearly half of test charges (47%) were priced between $100 – $199, and one in five (20%) were priced above $300.
The price of diagnostic tests can vary based on the type of test performed, where it is processed, the manufacturer, and CDC affiliation. We found hospital prices for laboratory tests developed by the CDC were generally lower than for other non-CDC tests, and tests utilizing high throughput technology – allowing for faster analysis of samples – were priced higher. Since we conducted this analysis in 2020, there has been an increase in the number of different COVID diagnostic tests, particularly respiratory panels that test for COVID as well as other viruses. For example, respiratory panels that include detection for SARS-CoV-2 and the flu tend to be priced more highly than those that just test for the coronavirus.
Self-pay rates. While there are federal and state resources that cover the cost of COVID testing for many uninsured patients, they may still face costs or be billed for care in certain circumstances. Thirty-nine hospitals listed the exact discounted rate or price of COVID-19 diagnostic tests for self-pay individuals, which ranged between $32 to $478 per test. A number of additional hospitals indicated on their websites that uninsured or self-pay individuals who didn’t qualify for governmental programs such as Medicaid could receive free or discounted care through hospital financial assistance programs. Patients receiving discounts would be charged for a portion of the listed price (e.g. 75% or 50% of the list price), though finding the exact amount of the discount can be challenging and often entails contacting the hospital to apply for discounted care.
Related costs. Aside from the cost of a diagnostic test itself, hospitals also bill for specimen collection and the visit itself, whether in-person or through telemedicine. In most cases, the total cost of care for a test and its related services was not easily distinguishable on hospital websites. However, 30 hospitals clearly posted the list price for specimen collection, which ranged from $18- $240. One hospital listed three different charges based on where the specimen was collected – in a skilled nursing facility, at home, or an outpatient setting.
For self-pay individuals, the cost of screening as well as testing may add up. In our review in 2020, one hospital’s price estimator tool demonstrated that the price of COVID-19 screening would be $77 before discounts, and $31 after applying a 60% discount for qualifying self-pay individuals. Such charges in addition to the cost of a diagnostic test and specimen collection can be financially burdensome or prohibitive for uninsured or self-pay patients, especially given the current labor market and high rates of unemployment.
Reasons why insured patients seeking a COVID-19 test might receive bills
With the FFCRA and the CARES Act, Congress signaled that it wants to eliminate cost barriers for COVID-19 testing for patients, whether they are insured or not. While the Biden Administration has clarified that all testing conducted for diagnostic purposes must be covered – including testing for asymptomatic people and without requiring medical screenings – there are still cases in which patients may be billed for tests; for example, if they are billed directly from an out-of-network provider. Some patients still face out-of-pocket costs related to COVID-19 testing, which our analysis shows can be high and unpredictable.
Patients who seek a test, but don’t receive one, may still be billed for COVID-19 test-related services. Federal law requires health plans to cover visits (and other related services) that result in the administration of COVID-19 testing with no cost sharing. News reports have described insured patients who faced medical bills after they sought COVID-19 testing but were not given a test. For example, one patient experiencing symptoms sought emergency care but was not tested because the facility did not have COVID-19 test kits available. Her health plan applied the deductible to the emergency visit, leaving the patient to pay more than $1,800.
For out-of-network COVID-19 tests, private plans are required to pay the full price that is posted online. However, federal law is silent with respect to the amount a health plan must pay for out-of-network testing and related services if the provider has not posted a cash price online and if the health plan and the provider cannot agree on a negotiated rate. Guidance issued by the federal government notes only that the out-of-network provider who fails to post prices online may be liable for a civil money penalty of up to $300 per day. One news story described an insured patient who faced high out-of-pocket costs after she received COVID-19 testing from an out-of-network free-standing emergency room that does not appear to have posted prices for COVID-19 testing services online.
Federal law does not require out-of-network providers to bill insurance directly for COVID-19 tests. Typically, providers that participate in a patient’s health plan network are contracted to bill that patient’s insurance directly and collect no more than in-network cost sharing directly from the patient. While the law requires coverage of out-of-network testing without cost sharing, patients who seek COVID-19 testing out-of-network may be required to pay up front for services, then submit claims to their health plan for reimbursement. Patients who don’t know how to manually submit out-of-network claims or who are too sick to do so may not succeed.
Federal coverage requirements apply only to individualized diagnostic testing, but not when conducted as part of employee “return to work” programs. Guidance issued by federal agencies states that health plans “are not required to provide coverage of testing such as for public health surveillance or employment purposes. But there is also no prohibition or limitation on plans and issuers providing coverage for such tests.” To the extent employers may expect or require their workers to be tested periodically for COVID-19, private health plans would not be required to cover such testing costs. However, employers that do require periodic testing for workers could choose to cover the cost of testing for their employees.
Federal requirements to cover COVID-19 tests do not apply to certain private plans. The FFCRA and CARES Act coverage requirements do not apply to non-ACA compliant plans, such as short-term health insurance policies. Some health insurers and short-term policy issuers have said they will voluntarily waive cost-sharing for COVID-19 tests under all of their policies.
Discussion
While the FFCRA and the CARES Act attempted to reduce COVID-19 testing costs for consumers, the cost and growing volume of testing could affect the cost of insurance overall. The list prices posted by 93 hospitals for COVID-19 tests vary widely and, in most cases, are significantly higher than the amount Medicare allows. Some patients might not be fully protected from receiving bills for COVID-19 testing or testing-related services. Patients in some circumstances may incur charges for related services, including the visit (in-person or telehealth) during which the patient is evaluated, the collection of test specimen, and other diagnostic tests or procedures ordered to confirm COVID-19 or rule out other possible conditions. These prices can also be high and unpredictable. Further complicating any effort to identify or categorize prices, the charges for COVID-19 testing and related services may be in a bundle, or the patient may receive separate bills from the hospital or testing facility, the attending health professional, the laboratory, and other providers involved in care. Unexpected bills for some or all of the charges can place significant financial strain on individuals and their families, and force patients to make difficult decisions about whether to pay for care or other household expenses.
These issues with health care costs are not new. The prices of medical care have been notoriously opaque in the U.S. Both insured and uninsured patients often only find out the full cost after they’ve received bills for their care. Hospitals and insurers have historically kept their negotiated rates secret, allowing the same provider to charge different rates for the same service. In an attempt to make the cost of care more transparent for patients and increase competition among providers and insurers, CMS released a final rule in November 2019 requiring hospitals to make public their standard charges, discounted charges, and insurer-negotiated rates for common services, effective January 2021. While price transparency in healthcare is not a panacea to high costs, implementation are also preventing patients and others from accessing full information on hospital prices.
Methods
We searched the websites of 102 different non-governmental adult hospitals for 50 states plus Washington D.C. to collect prices for COVID-19 diagnostic and antibody tests. The top two largest hospitals were defined as those with the most number of hospital beds, using data from the 2018 American Healthcare Association Annual Survey of Hospitals. These 102 hospitals represent 12.5% of total non-governmental hospitals beds in the U.S. In some states, these hospitals comprise a much greater share of total hospitals beds in that state. Some hospitals in the sample belong to larger health systems. In the five states where the top two largest hospitals were owned by the same health system, the hospitals had the same price for COVID-19 tests. In these states, we examined the prices of the third largest hospital in the state.
To find list prices, we systematically examined hospital websites between April 20 – 27, 2021 for this update. In 2020, prices for COVID tests were collected between June 15 – June 22, 2020.
We searched their billing and financials pages for sub-pages on price transparency or standard hospital charges. Many hospital websites had pages on price transparency in accordance with CMS rules on price transparency. When available, chargemasters or documents containing list prices of COVID-19 and/or other medical services were downloaded and then searched for COVID-19 tests. The chargemasters were searched using the terms “covid”, “sars”, “cov” and “coronavirus”. Some websites had price estimator tools, and those tools were also utilized to attempt to find COVID-19 test prices, but these often did not yield results. For these tools, we used the search terms “covid”, “sars”, “cov”, “coronavirus” along with the CPT codes “87635”, “87636”, “86318”, “86769”, and HCPCS codes “u0002’, “u0003”, “u0004”, “u00002”, and “u00003” when applicable.
For websites that didn’t have downloadable chargemasters, or for websites in which downloadable chargemasters did not contain price information for COVID-19 tests, we examined the remainder of the billing and financial information webpages, COVID-19 information webpages, and other related pages in “patients and visitors” sections of the hospital website. Finally, we searched the hospital website for price information using the terms including “price transparency”, “COVID test”, “COVID test cost”, “COVID test price”, “chargemaster” and “standard charges”. If this exhaustive search did not yield results, the hospital was coded as not having COVID-19 price information. While some hospitals indicated that price information could be requested through phone, email, or request forms, we did not contact hospitals for this information since the CARES Act required public posting of this information.
COVID-19 tests were identified by name or by code. Costs for specimen collection were only included if they were easily identifiable or grouped with COVID-19 test prices. When hospitals indicated discount rates or different prices for self-pay individuals, the separate price was included in our sample. When hospitals clearly listed discounts for self-pay as related to COVID-19 testing, those prices were calculated accordingly and collected.
The descriptions for the tests were often written in a medical billing shorthand and not standardized across all hospitals. We included COVID test prices for CPT codes 87635, 87636, 87637, 87811, 87426, and HCPCS U0001, U0002, U0003, U0004, but did not include CPT 0202u.
The Peterson Center on Healthcare and KFF are partnering to monitor how well the U.S. healthcare system is performing in terms of quality and cost.